Industries
Interventional
Interventional catheters integrated with cables and sensors are minimally invasive and produce high resolution images. The combination of connectivity and sensor solutions with Creganna Medical and AdvancedCath’s expertise in the medical device market provides customers with an unmatched set of capabilities and technologies.
TE has a long history of providing interconnect solutions for the medical industry. The acquisitions of Creganna Medical and AdvancedCath expand TE’s Medical business and positions us to be a leading provider of connectivity and sensor solutions in the high-growth medical device market. In conjunction with our industry-leading fine wire manufacturing, we support the medical devices trend toward minimally invasive catheter solutions. The custom design and manufacturing of advanced catheter systems, guidewires, medical balloons, extrusions, and laser micro-machined and welded assemblies uses best-in-class technologies with industry-leading expertise in lifesaving therapy areas. Key markets served include peripheral vascular, structural heart, electrophysiology and neurovascular. We partner with you to innovate smaller catheters that carry more signals, increasing functionality and improving workflow. We can now offer customers an unmatched combination of sensor, material science, electrical and biomedical technologies and a single source of manufacturing expertise for advanced medical devices.
Now Part of the TE Family
Creganna Medical & AdvancedCath
Creganna Medical is ranked among the Top 10 global companies in the medical device outsourcing industry and specializes in the design and manufacture of minimally invasive delivery and access devices for a range of therapies. Serving over 400 companies across 30 countries, Creganna Medical’s customers include the world’s leading medical device and life science companies. Headquartered in Galway, Ireland, Creganna Medical has a global operational footprint with design and manufacturing facilities spanning the US, South America, Europe and Asia.
AdvancedCath
AdvancedCath is a leading player in the design, development and manufacturing of advanced catheter systems. AdvancedCath works with medical device OEMs to design and manufacture catheter and guidewire systems used in complex interventional and endosurgical procedures.
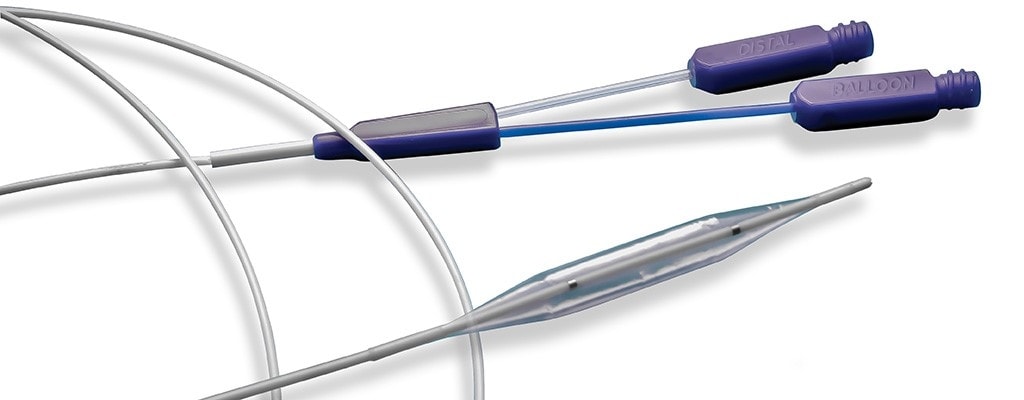
High-Level Catheter Assemblies
Choose TE as your catheter assembly development partner to leverage system-level design integration and engineering expertise in braiding, reflow, shaft assembly, electrodes/marker band attachments, steering wire integration and other value-add catheter assemblies.

Fine Wire Cable Assemblies
With coax diameters as small as 0.17mm and termination pitches as small as 100 microns, our fine wire capabilities can help you increase sensor density, combine measurement and therapy, and integrate real-time visualization in your catheter device.
Fine Wire Catheter Assemblies
TE has engineering and manufacturing expertise in catheter shaft assembly, component molding, cable assembly and connector design. By increasing the level of feature integration, we can enhance catheter function without increasing catheter size.
Custom Connectors
Our catheter connectors deliver signal fidelity, reliable connectivity and contact density, while addressing ease-of-use, clinical workflow and cost requirements.
Molded and Overmolded Parts and Assemblies
Our molded components deliver light, ergonomic and user friendly control interfaces for your catheter handpiece. We can overmold contacts and leads to generate custom switches and controls and work with you to select the appropriate molding material for your devices.
Medical Grade Heat Shrink Tubing
-
Heat Shrink Tubing
TE's Altera line of medical-grade heat-shrink tubing can be used as an aid to delicate catheter assembly processes. TE’s expertise in material formulation and extrusion, irradiation, and expansion can provide you with the tubing to meet your specifications.

Benefits
- Catheters and value-add catheter assemblies with dense, compact packaging of diverse content— electrical, fiber, steering, fluid and more
- Specialized in ultra-fine wire and microscopic, high-density termination
- Manufacturing automation
- Rigorous medical device quality systems
- Design for cost and manufacturing
- Nimble product development enables accelerated go-to-market
With TE Medical, you get more than just a components supplier. You get an integrated solutions partner who can develop your idea from design to prototype to manufacturing and assembly. The value we provide —engineering expertise, extensive manufacturing capabilities, global footprint, and focus on the end user— helps you compete.